DoriVac: DNA Origami Vaccines Could Replace mRNA
Harvard’s DoriVac vaccine platform uses folded DNA nanostructures to deliver immune responses comparable to mRNA vaccines while being stable without cold storage.
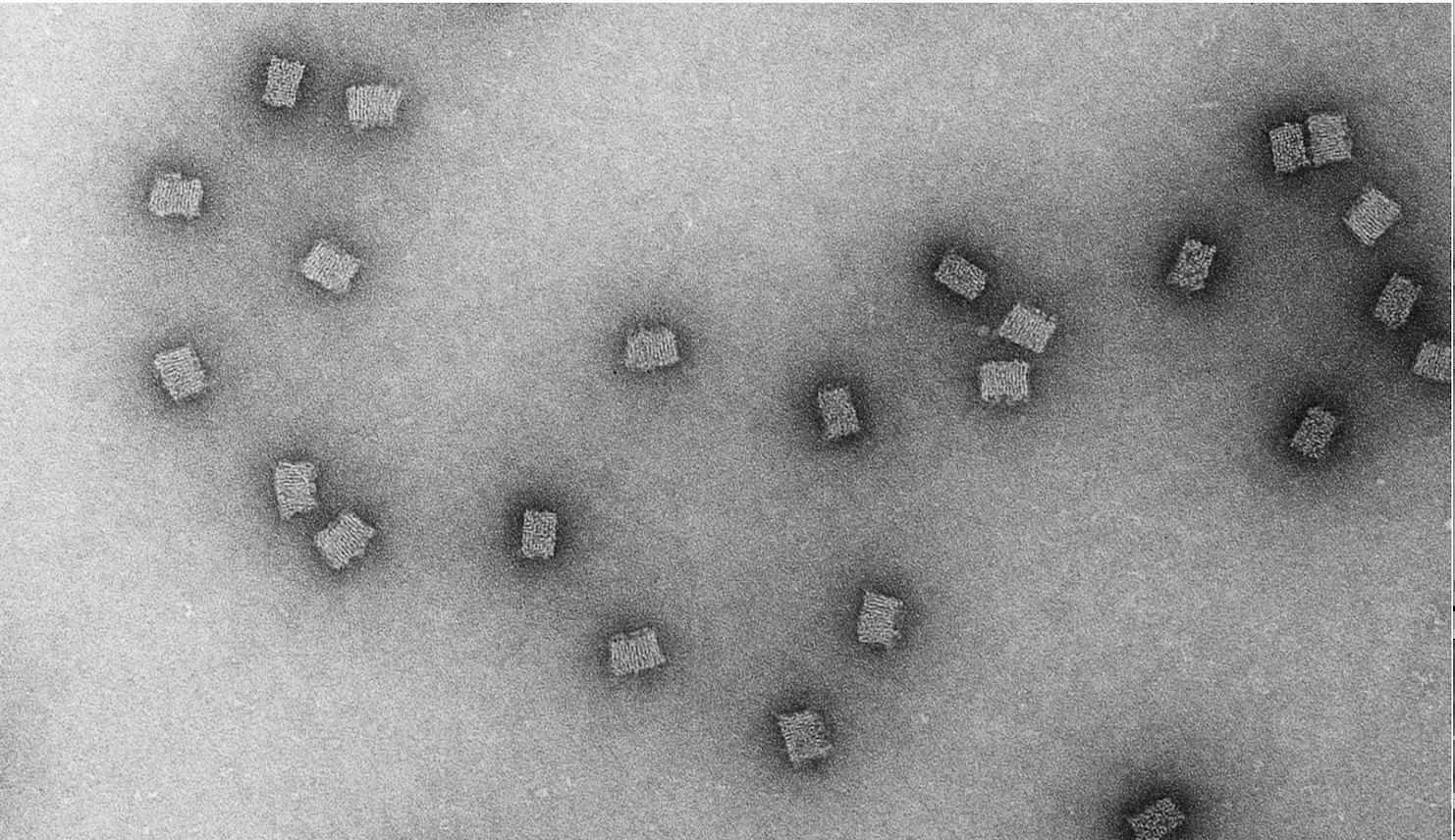
What Is DoriVac?
DoriVac is a next-generation vaccine platform developed by researchers at Harvard University’s Wyss Institute. Instead of using mRNA to instruct cells to produce viral proteins like the Pfizer or Moderna vaccines, DoriVac uses DNA origami technology to fold a long strand of DNA into a square-block nanostructure approximately 50 nanometers in size.
This nanostructure functions as a smart vaccine scaffold: one face of the square block carries disease-specific antigens, while the other face carries adjuvants that stimulate the immune system. This precise spatial arrangement allows unprecedented control over the immune response.
The research, published in March 2026 in Nature Biomedical Engineering, demonstrates that DoriVac generates immune responses comparable to mRNA vaccines in preclinical studies while addressing one of mRNA vaccines’ biggest challenges: the requirement for strict cold-chain storage.
How It Works
The DoriVac process begins with a long strand of DNA engineered to self-fold into a specific shape when mixed with hundreds of short complementary DNA strands called staples. This self-assembly process occurs in a test tube without complex equipment, significantly reducing manufacturing costs.
The key innovation of DoriVac is its ability to target the conserved HR2 peptide region — a part of viruses that changes little across variants and different viral families. This means a single DoriVac vaccine could provide protection against multiple diseases, from COVID-19 to HIV and Ebola.
When injected into the body, DoriVac nanoparticles are recognized and processed by antigen-presenting cells, activating both innate and adaptive immune responses. The result is a strong and durable immune response without requiring cold storage.
Global Health Impact
One of the biggest barriers to global vaccine distribution is the cold-chain requirement. Pfizer’s mRNA vaccine requires storage at negative 70 degrees Celsius, while Moderna requires negative 20 degrees. This makes distributing vaccines to rural areas in developing countries extremely difficult and costly.
DoriVac could change this equation. With stability at room temperature, DNA origami vaccines could be shipped and stored anywhere without specialized refrigeration. This is particularly important for countries in Africa, Southeast Asia, and South America where cold storage infrastructure is limited.
Furthermore, DoriVac’s modular design allows rapid vaccine adjustment when new virus variants emerge. Instead of redesigning the entire mRNA sequence, researchers simply change the antigens attached to the nanoparticle surface — a significantly faster and simpler process.
References
- [1]DNA origami vaccines next leap beyond mRNAScienceDaily
- [2]Beyond mRNA: DNA Origami as Vaccine PlatformSciTechDaily
- [3]DoriVac TechnologyWyss Institute
- [4]DNA Origami: the future of vaccines -- Nature Reviews, March 2026Nature Reviews
▸ mRNA vaccines require -70C storage -- DoriVac is stable at room temperature, potentially saving millions in rural Southeast Asia where specialized freezers don't exist.
▸ If DoriVac succeeds in clinical trials, vaccine production costs could drop 60-80% -- helping developing nations achieve vaccine independence faster.
Related: Sleep Optimization and Neurowellness Trend.
Frequently Asked Questions
The most common questions about DoriVac vaccine technology and DNA origami.
Related Topics
Stay on top of trends
Bookmark this page and check back often for the latest updates and insights.